Pro-curo gives tissue establishments, tissue banks, and HTA-licensed organisations a single platform to manage donor-to-recipient traceability, track samples through every stage, and maintain the audit trails required by the Human Tissue Act and the EU Tissue Directive — securely in the cloud or on your own infrastructure.
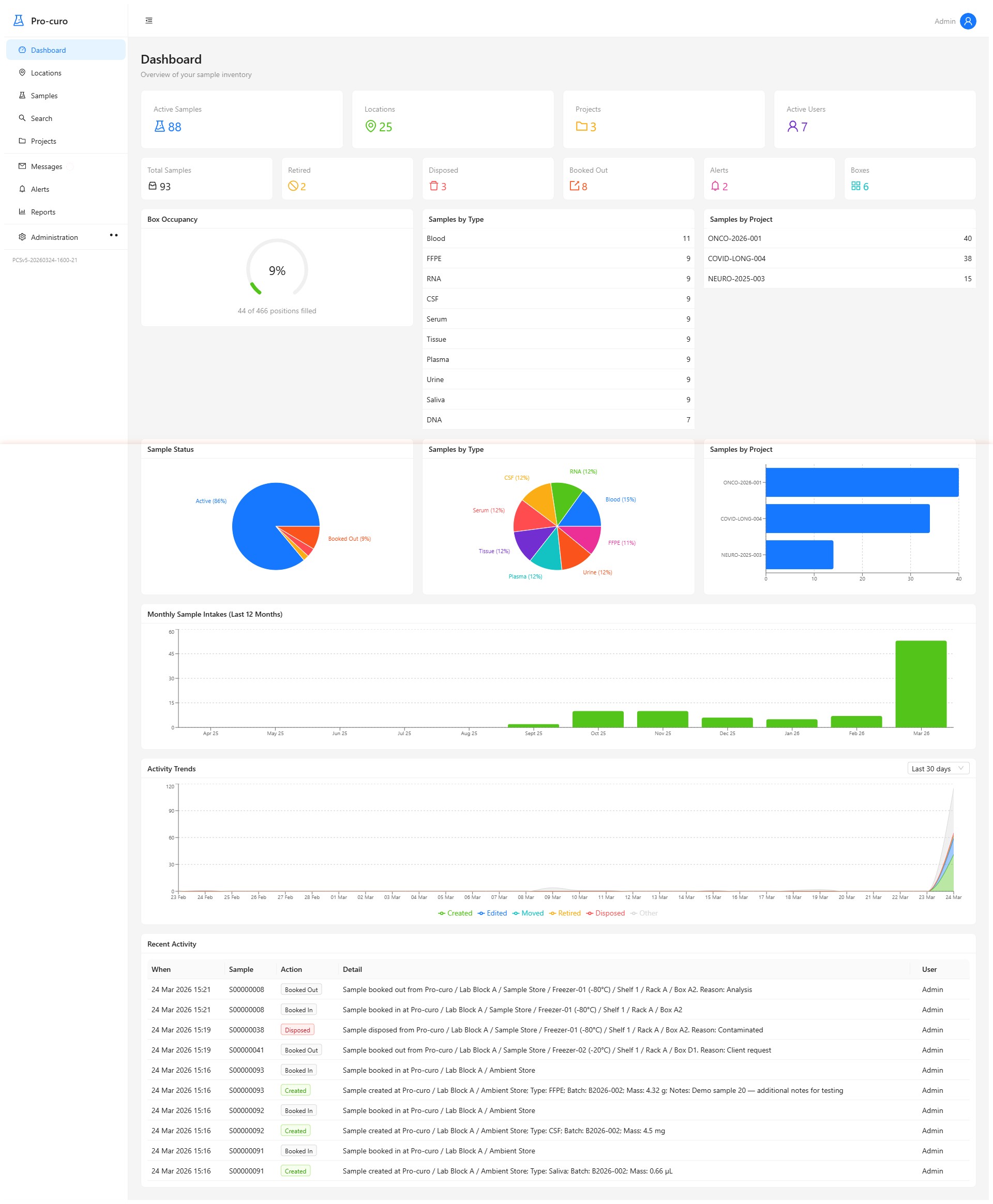
Pro-curo combines rigorous traceability with a clean, intuitive interface — so your team can focus on processing, quality, and patient safety rather than battling spreadsheets and paper records.
Access your data from any device with a modern browser — no installation required. Works on desktops, tablets, and mobile.
Deploy on our secure cloud infrastructure or host on your own servers. Your data, your rules — we support both.
Role-based access control, JWT authentication, and a full audit trail on every record. Built for regulated industries.
Real-time KPIs, charts, and project overviews at a glance. Make informed decisions without digging through reports.
From donor procurement to recipient distribution, Pro-curo covers every stage required by the Human Tissue Act and EU Tissue Directive.
Track tissues and cells through every stage — donation, procurement, testing, processing, preservation, storage, and distribution. Maintain full donor-to-recipient traceability with consent records, labelling, and chain-of-custody documentation. Find any sample in seconds with powerful search and filtering.
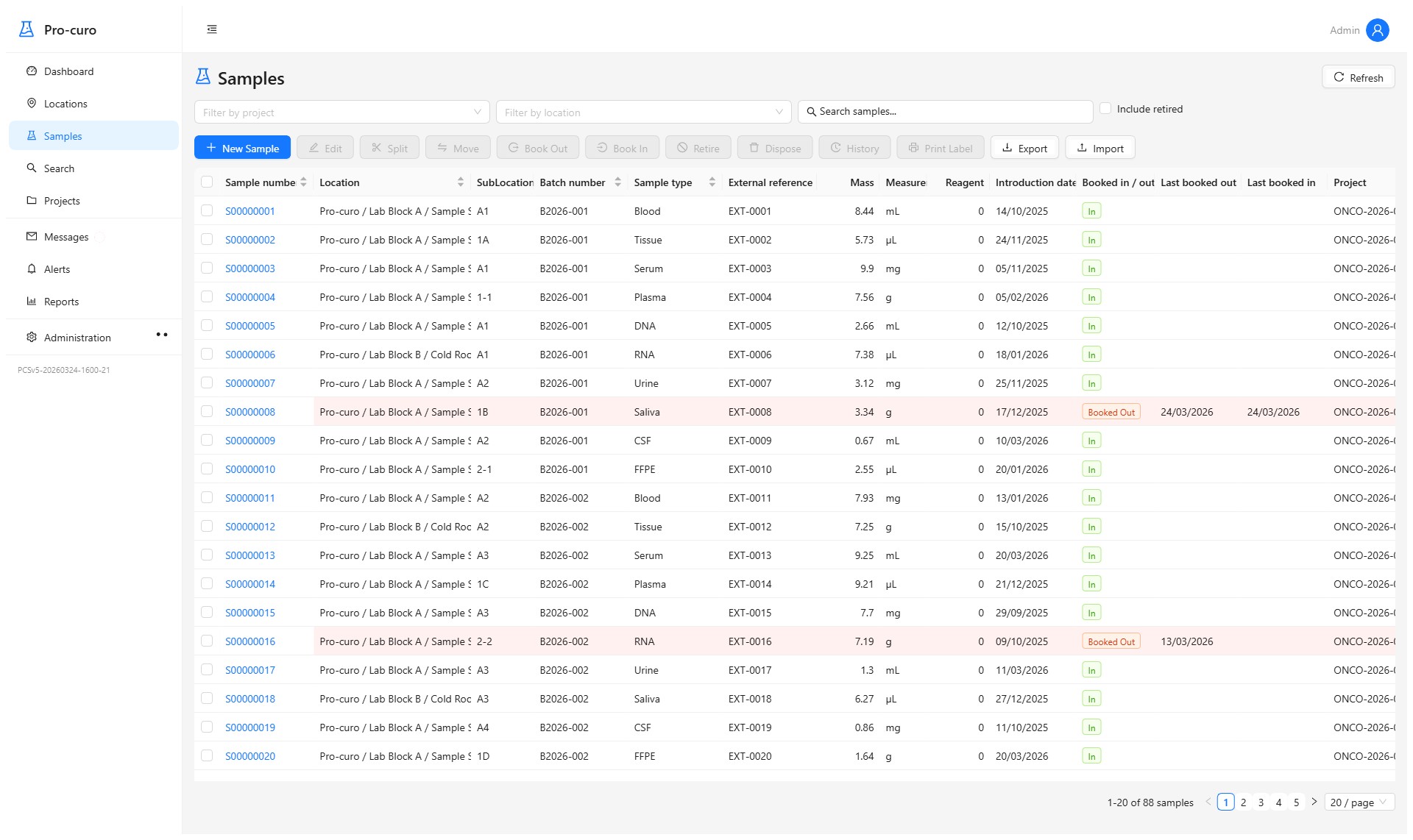
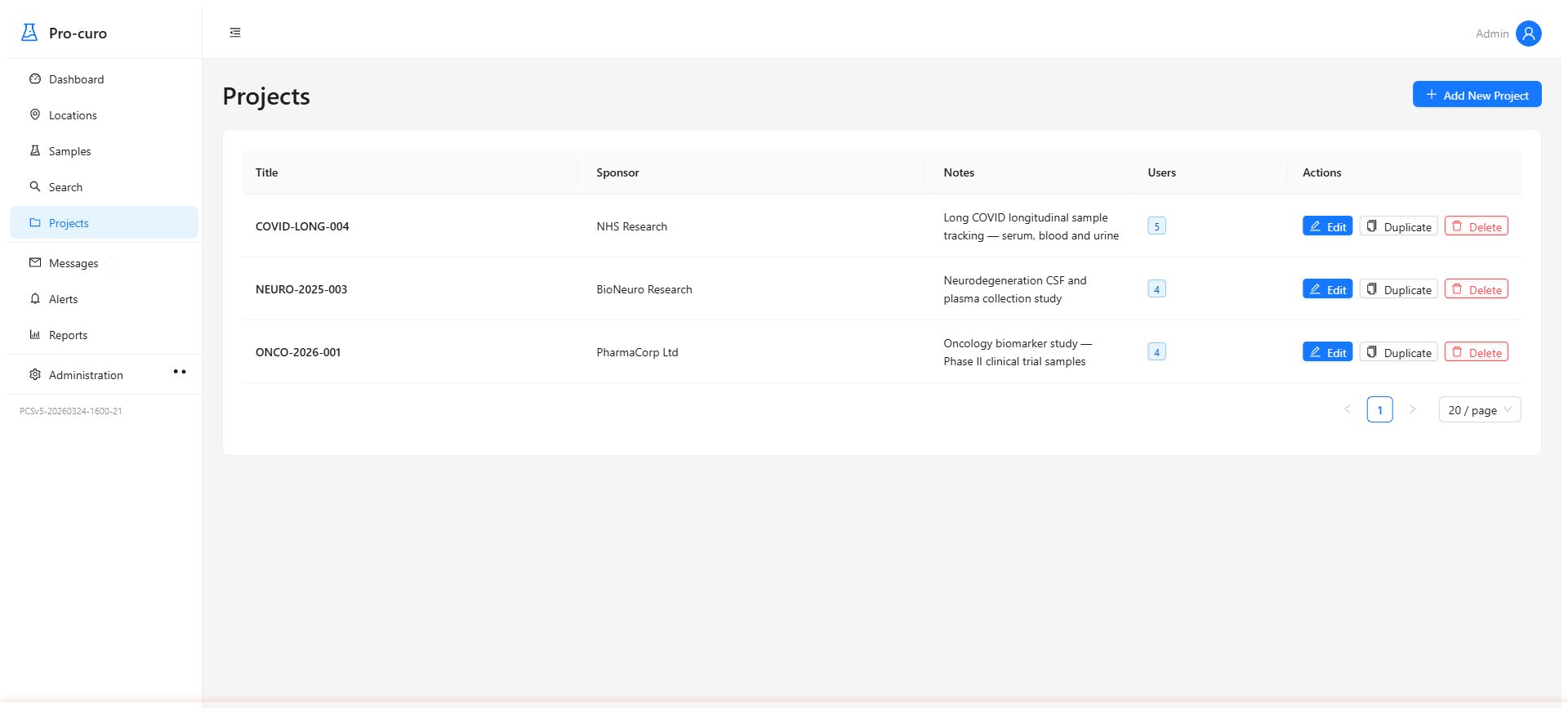
Organise work by project, donor, or establishment. Track processing workflows, manage quality control steps, and keep everything linked to the right tissues and locations. Replace paper-based systems with structured, auditable workflows.
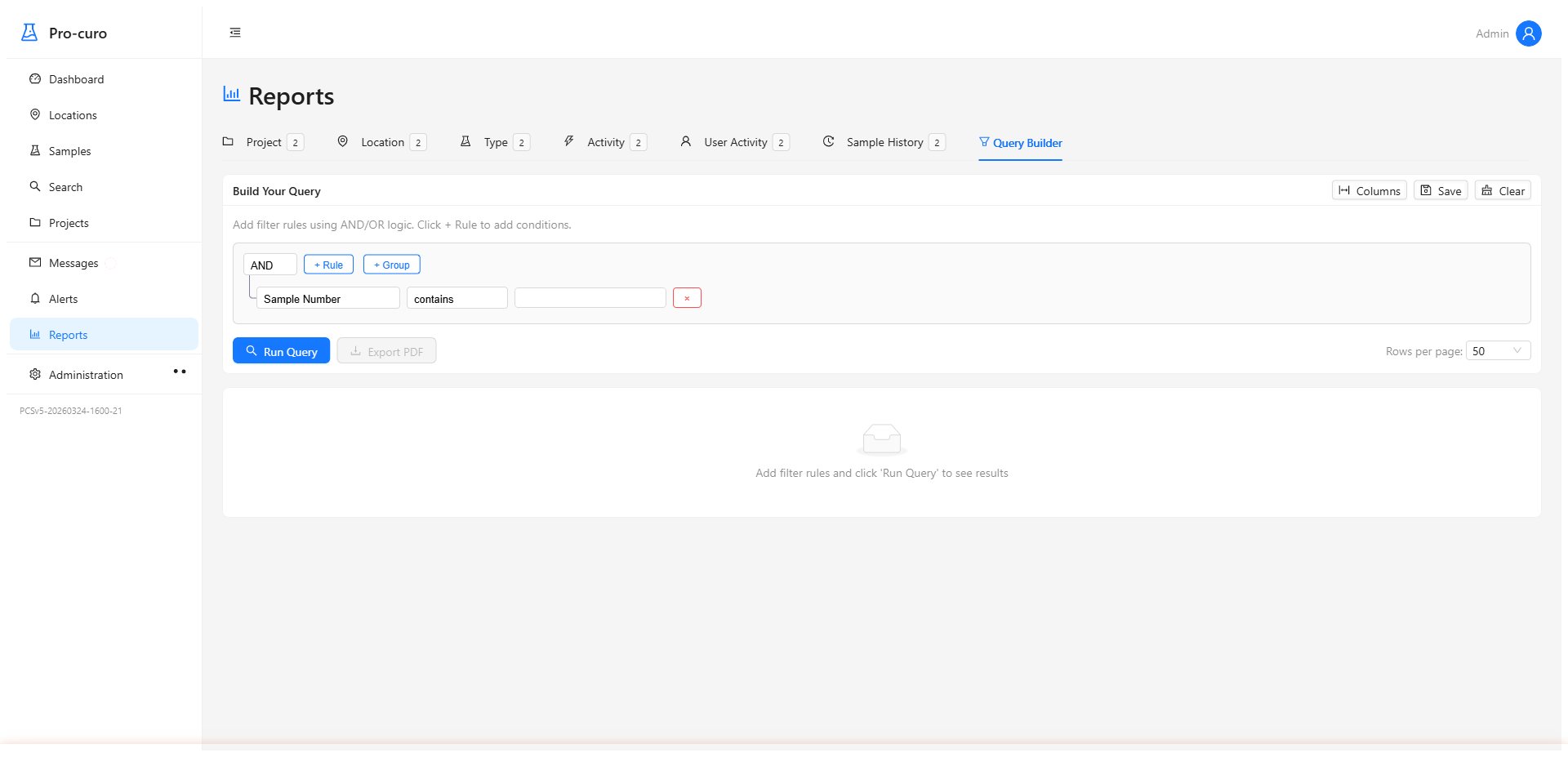
Build custom queries with a visual drag-and-drop builder — no SQL needed. Generate the reports your HTA inspector needs, export to CSV, create PDF reports, and schedule recurring reports for your team, management, or the competent authority.
Pro-curo was purpose-built to meet the traceability requirements of the Human Tissue Act 2004 and the EU Tissue Directive (2004/23/EC). We help tissue establishments maintain full donor-to-recipient traceability, manage quality systems, and stay inspection-ready at all times.
Our core client base. Pro-curo provides tissue banks with end-to-end traceability — from donor procurement through processing, storage, quarantine, and distribution to recipients.
Track corneal and ocular tissue from donation through evaluation, processing, storage, and distribution. Maintain the detailed records required for HTA licensing and inspection.
Manage bone, tendon, cartilage, and other musculoskeletal tissue through every stage — procurement, processing, irradiation, storage, and onward distribution to surgical teams.
Pro-curo supports NHS tissue services and hospital-based tissue programmes with the structured workflows and audit trails needed for HTA compliance and internal governance.
Universities, research institutions, and anatomy departments use Pro-curo to manage consent records, track tissue samples, and maintain the documentation required under the Human Tissue Act.
The EU Tissue Directive applies across all member states. Pro-curo supports tissue establishments internationally with multilingual traceability and compliance with both EU and national regulations.
Explore our white papers on tissue traceability, read case studies from organisations like yours, and stay current with industry developments.
From small research groups to multi-site NHS biobanks — see how Pro-curo is used in practice across the UK.
View Case StudiesIn-depth guides on HTA compliance, chain of custody, the EU Tissue Directive, and moving from paper to electronic systems.
Browse White PapersCurated news from laboratory, life science, and tissue banking sources relevant to HTA-regulated organisations.
Read Industry NewsBook a personalised demo and discover how Pro-curo can streamline your sample and project management.
Request a Demo